How Real-World Evidence is Elevating Healthcare Standards in India?
RWE in India: Advancing Post-Market Surveillance
India’s healthcare landscape has increasingly recognized the importance of Real-World Evidence (RWE) studies. As the Clinical Research Organization in India continues to grow, ensuring that medical products—both drugs and devices—meet safety and efficacy standards in the real world is becoming critical. RWE studies play a vital role in achieving this by providing essential insights once products are on the market. As a Top Clinical Research Organization in India, Abiogenesis Clinpharm is committed to leading the way in utilizing RWE to enhance patient safety and treatment effectiveness.
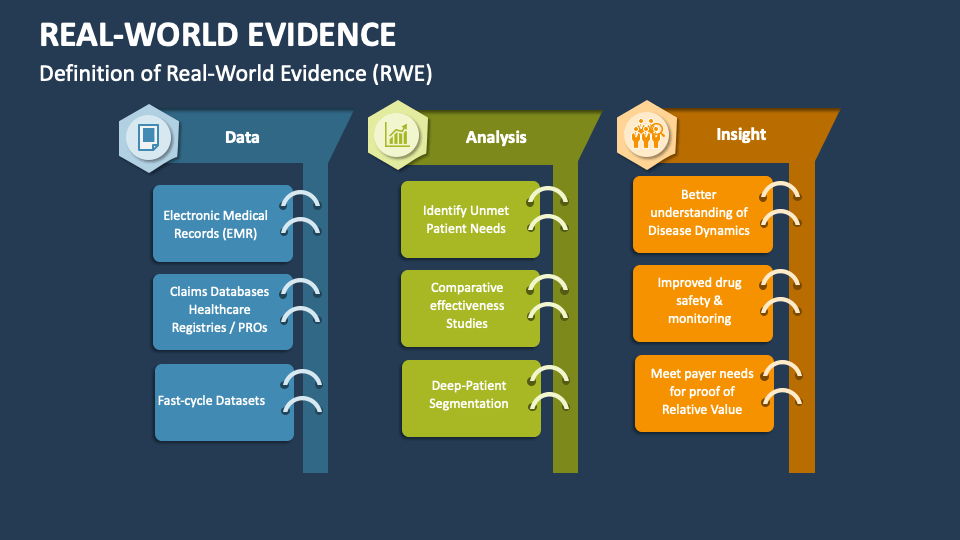
What is Real-World Evidence (RWE)?
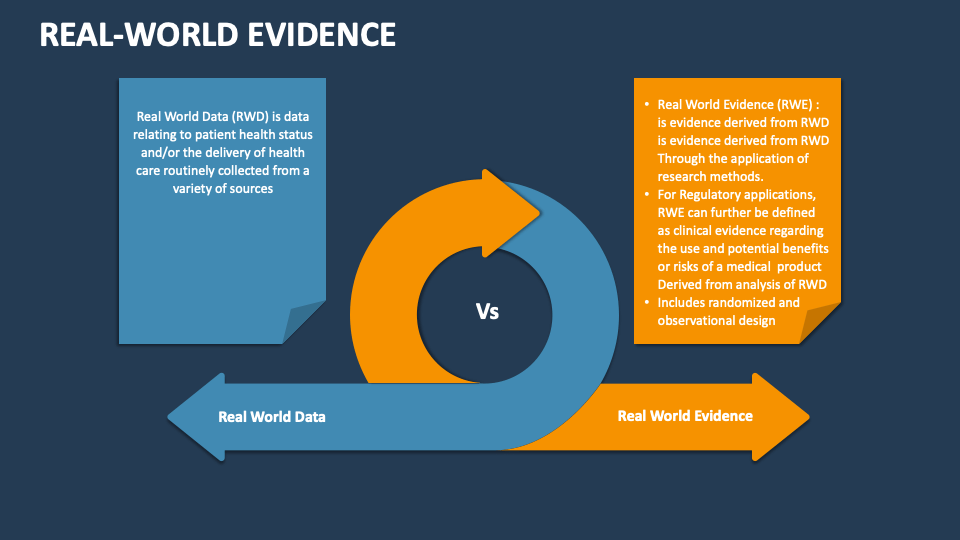
RWE refers to data collected outside of controlled clinical trials, capturing the actual usage of medical products. This data is sourced from electronic health records (EHRs), patient registries, insurance claims, and wearable devices. By tracking long-term effectiveness and safety, RWE helps us understand how treatments perform across diverse patient populations, extending beyond the scope of pre-market studies. As a Top Clinical Research Organization in India, we leverage RWE to deliver comprehensive, real-world insights that drive better healthcare outcomes. Our expertise enables us to collect, analyze, and interpret real-world data, supporting healthcare providers, regulatory bodies, and pharmaceutical companies in making informed decisions. This collaborative approach not only improves patient safety but also ensures treatments remain effective and relevant to real-world needs.
RWE also offers valuable insights into the effectiveness of treatments in real-world scenarios, highlighting potential safety concerns and long-term benefits. By understanding patient behaviors, treatment adherence, and the impact of environmental and genetic factors, we provide data-driven solutions to address the unique healthcare challenges in India. Our approach enables healthcare providers to optimize patient care, tailor therapies, and achieve better clinical outcomes, while pharmaceutical companies gain a deeper understanding of their products’ performance beyond clinical trials. As a Clinical Research Organization in India, Abiogenesis Clinpharm remains committed to advancing healthcare through RWE, delivering meaningful, actionable insights for improved patient outcomes.
The Role of Real-World Evidence in Meeting Post-Market Regulatory Requirements
In India, Post-Market Surveillance (PMS) is crucial due to the country’s large and diverse patient population, which may not be fully represented in clinical trials. While drugs and devices are evaluated for efficacy during trials, their performance can differ in real-world scenarios due to factors such as genetic variations, co-morbidities, and environmental influences.
As a top clinical research organization in India, we understand the importance of effective PMS strategies. Regulatory bodies like the Central Drugs Standard Control Organization (CDSCO) are increasingly using Real-World Evidence (RWE) to support safety evaluations and effectiveness assessments. By naturally integrating RWE into PMS, companies can identify safety signals, monitor long-term outcomes, and ensure that their products meet both regulatory requirements and patient expectations.
Key contributions of RWE include:
Enhancing Patient Safety RWE helps monitor adverse events and side effects that might not have appeared during clinical trials. This continuous monitoring allows healthcare providers and regulators to act swiftly and ensure patient safety, particularly in large populations.
Assessing Long-Term Effectiveness The real-world use of medical products can provide valuable insights into their long-term benefits and effectiveness. By tracking patients over extended periods, RWE helps assess how well a product works over time, especially for chronic diseases or long-term treatments.
Improving Regulatory Compliance For pharmaceutical companies and device manufacturers in India, adhering to post-market surveillance regulations is mandatory. RWE plays a key role in helping companies meet regulatory requirements by reporting safety concerns, adverse reactions, and the continued effectiveness of their products. Real-world data also aids in fulfilling the periodic reporting obligations set by authorities, ensuring ongoing compliance.
Identifying Unmet Medical Needs RWE provides valuable insights into areas where existing treatments might be inadequate, allowing regulators and healthcare providers to identify gaps in care. This can lead to the development of new therapies or modifications to existing treatments to better meet patient needs.
Collaborations for Collecting Real-World Data in India
Real-World Evidence is only as useful as the data collected, and gathering this data in India requires collaboration across various stakeholders, including healthcare providers, insurers, pharmaceutical companies, and even technology firms. As a top clinical research organization in India, we understand the importance of robust and accurate data collection. Our expertise in seamlessly coordinating with multiple stakeholders ensures reliable, high-quality real-world evidence that drives better healthcare decisions and innovative solutions.
Partnerships with Healthcare Providers
Hospitals and healthcare organizations are a critical part of the real-world data collection process. These institutions provide real-time data on patient outcomes, adverse reactions, and long-term treatment effects. By collaborating with healthcare providers, pharmaceutical companies can ensure that RWE accurately reflects how products perform in clinical practice.Collaboration with Insurance Providers
Insurance companies maintain extensive records of patient treatments, prescriptions, and outcomes. This data is invaluable for understanding how medical products perform in a larger, diverse population. Collaboration with insurance companies can provide a wealth of data that supplements clinical trial information and enhances post-market surveillance efforts.Leveraging Digital Health Tools
Digital health tools like mobile applications, wearable devices, and patient portals provide opportunities for real-time data collection. These tools enable patients to track their health, symptoms, and treatment responses on a daily basis, providing a continuous stream of real-world data. As a top clinical research organization in India, we leverage these digital tools to enhance the quality and accuracy of real-world evidence, enabling more informed decision-making and better patient outcomes.
If you want to know more about Top clinical Research Organization In India
Conclusion:
Abiogenesis Clinpharm, a Top Clinical Research Organization in India, is committed to enhancing healthcare outcomes by leveraging Real-World Evidence. Our collaborative approach ensures comprehensive data collection that supports regulatory submissions and provides valuable insights into patient needs, treatment responses, and long-term outcomes. As a Clinical Research Organization in Hyderabad, we continuously work toward delivering reliable, data-driven solutions that elevate healthcare standards and patient safety in India. As a leader in Top Clinical Research Organizations in India, we remain dedicated to advancing healthcare through RWE, ultimately improving the lives of patients across the nation.
This approach further solidifies our position as a Top Clinical Research Organization in India committed to driving better patient outcomes through Real-World Evidence, ensuring effective and personalized treatments across India. Additionally, our work as a Clinical Research Companies in India and clinical research companies in hyderabad ensures that we are at the forefront of providing tailored solutions to meet diverse patient needs in the real world.



